Learn more about the DoD perspective when awarding funding.
In order to understand the best strategies to gain Department of Defense (DoD) funding, you must first understand what constraints and limits the DoD staff has for grant writing. It can be surprisingly difficult to get money out the door, hence the unofficial tagline of DoD funding employees “It shouldn’t be this hard to give away money.”
🔬Learn more: What are SBIR/STTR Grants?
Most people think of the DoD as being focused on investing in products and services that the DoD is interested in using themselves, especially for military purposes. They are more often the customer or beneficiary of the technologies they choose to fund, as opposed to other organizations like the National Institutes of Health (NIH). Multiple military focused research programs are a part of the DoD (full list in graphic below).

But, despite their main focus being on military advancement and new technologies, the DoD also funds research for the general public good. A program called Congressionally Directed Medical Research Program (CDMRP) works on various topics for the public good, like research involving breast cancer, autism, or vision science. These topics are more similar to NIH funding than a specific military contract, but the DoD funds the CDMRP.
The CDMRP partly came about because of budgetary restrictions involving the NIH. The government wanted to fund more breast cancer research (and similar public problems), but the NIH’s budget would not allow for more funding. So, congress used the CDMRP to funnel more money for medical research using the DoD’s budgetary rules.
🔬Read more: Department of Defense (DoD) Funding: Understanding the Options
A DoD acquisition is the term for how the DoD acquires technology and products to address unmet needs, or ‘Requirements.’ DoD acquisitions can become very complicated and detailed, but this article will attempt to outline just the basics.
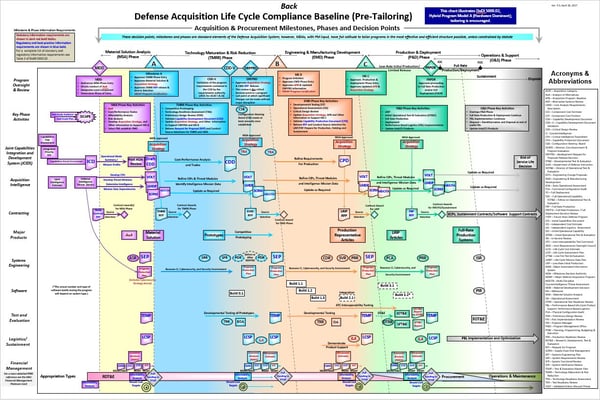
Above is an example of a wall chart that many DoD employees need to follow for defense acquisitions. As you can see the process is quite complicated; you are not expected to memorize this chart when working with the DoD.
A ‘Requirement’ as briefly mentioned earlier, is the product of a system that identifies, assesses, validates, and prioritizes needs across the military. The Joint Capabilities Integration and Development System (JCIDS) office governs these Requirements. The JCIDS office defines and locates the unmet needs in the military. Another process involves the funding of these projects and allocating resources, named the Planning, Programming, Budgeting, and Execution (PPBE) process.
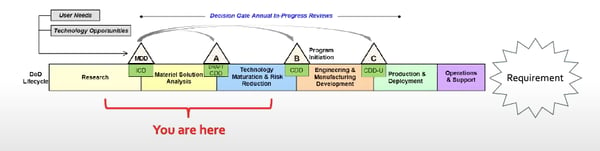
The life cycle is the DoD’s plan or process to fulfill the previously defined Requirements. First, the DoD researches if their problem/needs can be fixed with something that already exists. If not, they will make the decision to develop that technology themselves (or fund someone else to). Linear developments best suit this process, like an aircraft carrier (or something of the material sort). Something that would not work well with this linear process is software development, because it is constantly evolving and growing. This means that this traditional and linear framework might not work well with everyone’s technology. The example below is the most basic type of DoD life cycle
There is an ongoing attempt at the DoD to develop a framework that is less linear than the one above, but it is still in its early stages. There is also a different life cycle for pharmaceuticals and medical devices funded by the DoD. Check out the comparison below.
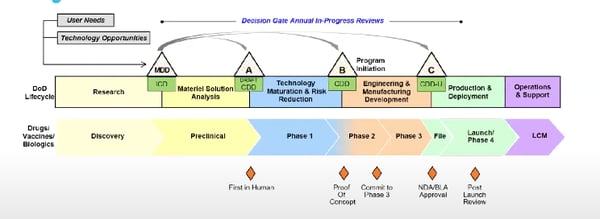
The life cycle for pharmaceuticals at the DoD.
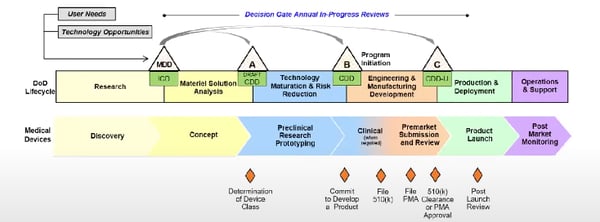
The life cycle for medical devices at the DoD.
🔬 Learn about: Funding Options for Biotech Startups
These life cycles are important because the DoD uses them to determine where you product is at along the pathway. They will assign you a number depending on what stage you are in, called Technology Readiness Levels (TRLs). The DoD wants to be able to see where your technology was at the start of their investment, and how your technology has progressed through the lifecycle after they have invested in it. For non-traditional technology the DoD can assign Knowledge Readiness Levels (KRLs), which are similar in nature.
Where your technology is along this lifecycle will affect what type of money the DoD can award you. There are specific ‘pots’ for each TRL that the DoD can pull money out of. That is why it is important for the DoD to have your technology identified and evaluated at a certain TRL, because they need to know what ‘pot’ they can allocate money from. Ultimately, the DoD staff looks to show their leadership that DoD money has been used to fulfill Requirements. If you understand how TRLs work and communicate with the DoD staff, you will be able to help them achieve their goals toward their specific Requirements and impress their leadership.
While DoD staffers are looking to maximize your funding and work toward DoD requirements, they are also trying to avoid breaking federal funding rules (of which there are many). There are general government funding rules that apply to the NIH, DoD, and National Science Foundation (NSF), as well as rules just for the DoD (more on that later).
All government appropriations need to follow the Anti-deficiency Act, the Misappropriation Act, and the Bona Fide Need Rule.
The Anti-deficiency Act was enacted “to prevent the incurring of obligations or the making of expenditures (outlays) in excess of amounts available in appropriations or funds.” This means that the DoD (or other agency) staff will not encourage you to do extra work beyond your approved reward with DoD money. You can only spend money on approved work.
The Misappropriation Act states that “funds appropriated by Congress be used only of the programs and purposes for which the appropriation was made.” This means that the DoD staff can not use funds for anything beyond their specific purpose. The Misappropriation Act makes it hard for the DoD to re-budget funds between different stages of development. An example of a violation of this act is using money set aside for prototyping for manufacturing a product instead.
The Bona Fide Need Rule “requires appropriated funds be used only for goods and services for which a need arises during the period of that appropriation’s availability for obligation.” The Bona Fide Need Rule means that you can not hold money that was allocated for a certain project for another time. This creates a time pressure for the DoD to spend money before it is sent back to the Treasury.
At the DoD there is also a difference between committed funds and obligated funds. Committed funds are ‘penciled in’ on the budget and recommended for funding. Obligated funds are a formal agreement to pay the awardee, who will usually receive an award letter. The DoD (and other agencies) are concerned with turning committed funds into obligated funds within their timeline. DoD staffers are concerned with their obligation rate to prove to their leadership that they have funded successful projects.
Staff at the DoD and other agencies are also concerned with making sure that researchers do not receive federal funding for the same work twice. Usually they will ask you about all of the other possible grants you may be receiving and have already received. This is where there is crossover between the NIH, DoD, and NSF. Each staff member wants to ensure that they are not funding a project that has already been funded by another agency. So, when drafting out proposals, make sure that your research objectives are specific and discrete. If you provide a broad overview on your first application, it will be harder to ask for funding again on the same project because they might overlap.
Many researches apply for multiple grants at the same time and can be overextended. Grant officers will also make sure that the researcher is able to give in maximum effort to each project.
🔬Related: How to Mine Federal Databases for Funding Opportunities
Now that we have covered the broad federal rules and concerns, we can learn more about DoD specific concerns.
One example of additional regulation from the DoD office would be their regulation of human and animal research. There are 2 offices that help regulate this: Human Research Protection Office (HRPO) and Animal Care and Use Review Office (ACURO). These offices act in addition to traditional Institutional Review Boards (IRBs). These offices will add delays to your research schedule (typically 2-3 months or longer). This is one of the reasons why it is next to impossible to carry out human subject research for a shorter award, like a 6 month SBIR award.
If you are looking to do research involving military populations there are a couple things you need to consider. It is typically considered a vulnerable population because there are concerns regarding consent within the command structure of the military. Typically, you will need the service member’s command to give permission for their participation. But, command is forbidden to be involved in recruitment for these studies. Service members and federal employees are also forbidden from receiving compensation for participating in a study.
These 3 issues can lead to issues with recruitment for military-based studies. They can lead DoD officers to be wary of studies that request research in military populations. In order for them to approve and fund your research, you need to work on convincing the DoD staff that you are prepared to interact with the military population. Having a letter of support from command and hiring recruitment staff with military experience is crucial.
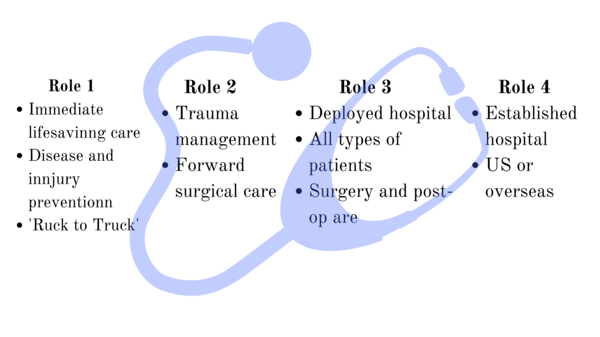
Most of the time the DoD is interested in rugged and lightweight products or devices if the indeed use is military based. In the example below for medical products you can see how there are different contexts for each product. Knowing the context that the DoD wants to apply your technology will help you understand how to design your product. For example, Role 1 care can’t be as heavy or delicate as Role 4 care in an established hospital.
Check out the resources below for more info on working with the DoD. Knowing the basics before contacting your assigned DoD staffer will help you ask more informed questions about your application.
Many of these are multi-page documents that can be hard to read all at once, but are great for referencing.
Generally in-person events are the best choice, but when not available you should contact the DoD office within your window of time that you are allotted. Find an excuse to talk to them so you can begin to develop that relationship.
The DoD is not able to control the SBIR grants as much because they are regulated by the Small Business Administration. The DoD will write the topic areas but are not as in control of the rules for an SBIR.
The grantors are trying to find new ways to work with the acquisition framework and rules to fit within a more lean iterative process. We can see this with their attempt to make a new acquisitions framework.
ScienceDocs will help you identify which agency is right for you, and the differences between them. They will also help to make sure that your technology fits what the agency is looking for, and for ultimate draft help or revisions. Click here if you want some more tips on a competitive SBIR/STTR proposal.
Not for CDMRP, because that is for general public use/benefit. For other DoD projects it will be specified if it is for military use only.
They are essentially second level IRBs. Once you have an approval from your IRB, you will need to send the office all of your information; this includes your informed consent documents, your protocols, your advertisement materials, etc. Then they will check it and add any DoD specific language they might need.
It depends on the end result and what the DoD asked for. However, asking for funding to complete a study in a military population and then transitioning to civilian mid-study is frowned upon.
Yes, at the CDMRP. The peer review process will produce a score. Programmatic review (which takes place after the first review) is a more subjective review of where it fits in the DoD portfolio. The score will depend on the funding solicitation that you apply to.
This content comes from a University Lab Partners webinar hosted in partnership with ScienceDocs, presented by Dr. Carly Kiselycznyk.
Dr. Kiselycznyk is a SBIR Consultant at ScienceDocs. She has written and negotiated funding opportunities for medical research and development as a DoD grant writer. She also was a point of contact for research scientists trying to navigate the priorities and regulations of DoD leadership
Be sure to subscribe to the ULP Youtube Channel to never miss another webinar, and connect with us on LinkedIn to stay in the loop!
Download The Ultimate Guide to Wet Lab Incubators in Southern California, a handbook to assist life science start-ups through the entire decision-making process to find wet lab space.
Download Now